DISK DIFFUSION
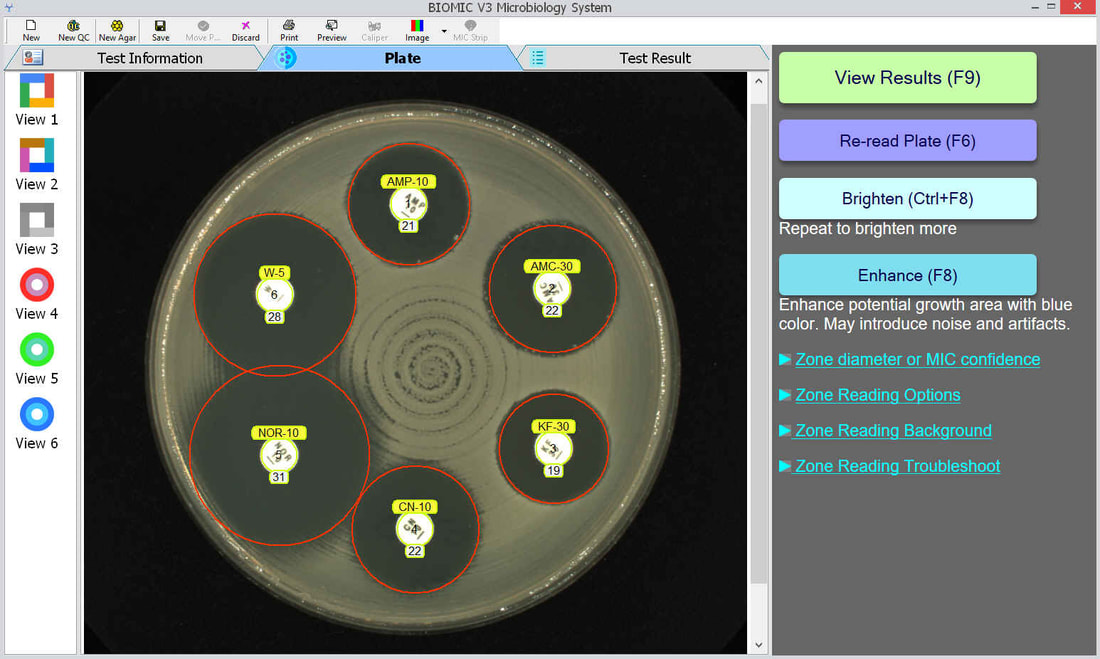
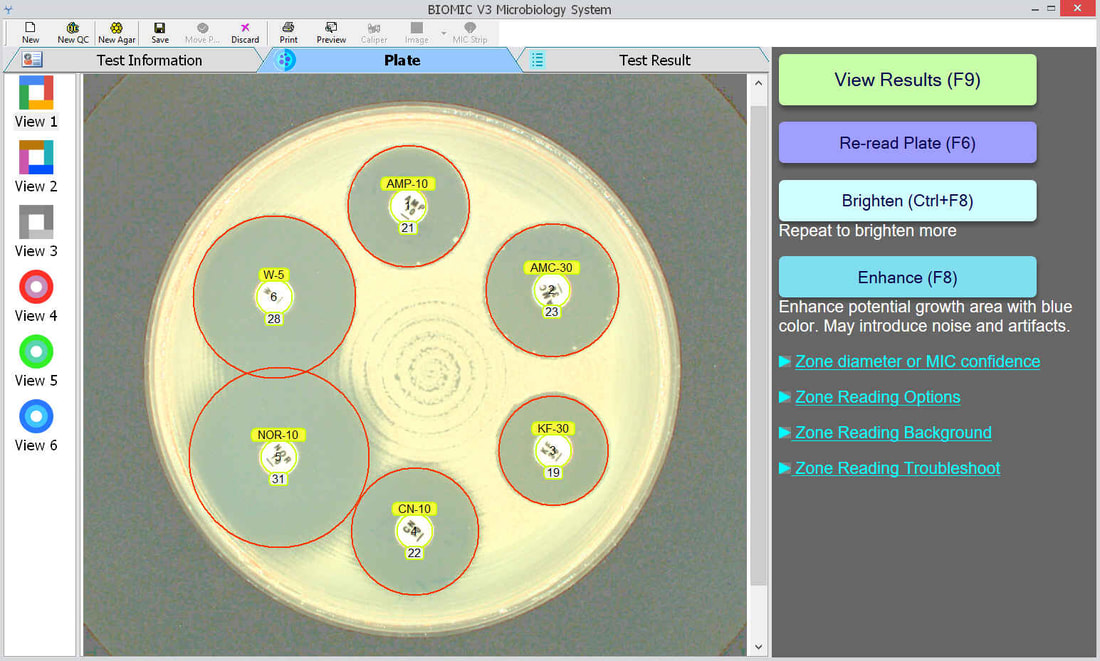
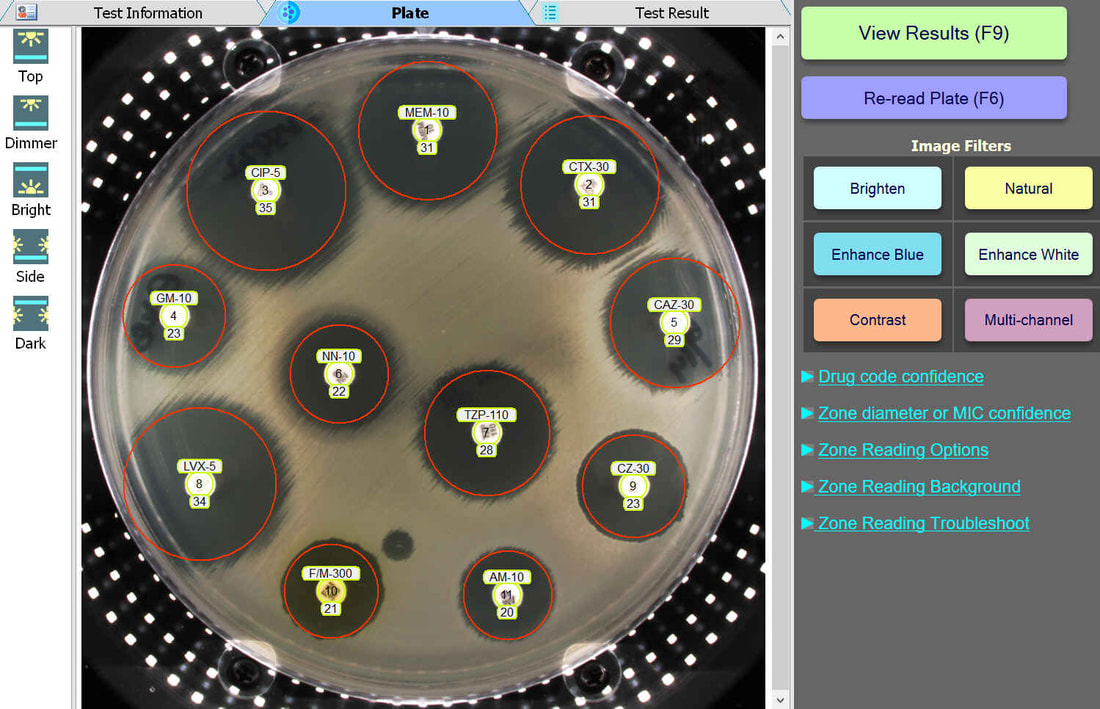
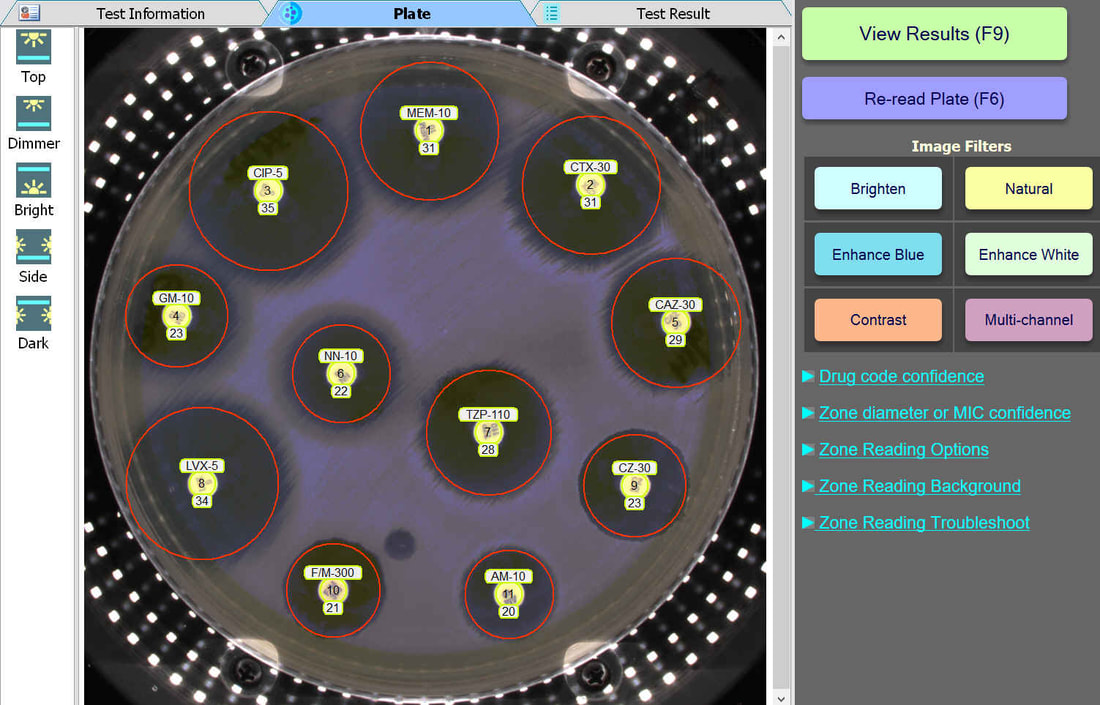
The BIOMIC V3 Microbiology System provides automated reading, interpreting, and expert review of CLSI or EUCAST antibiotic disk diffusion (Kirby-Bauer) tests on 90-150mm plates. This greatly improves accuracy, speed, and standardization of testing. AI algorithms generate test results and assist user decision making and reporting including pattern matching, character recognition, and image analysis.
|
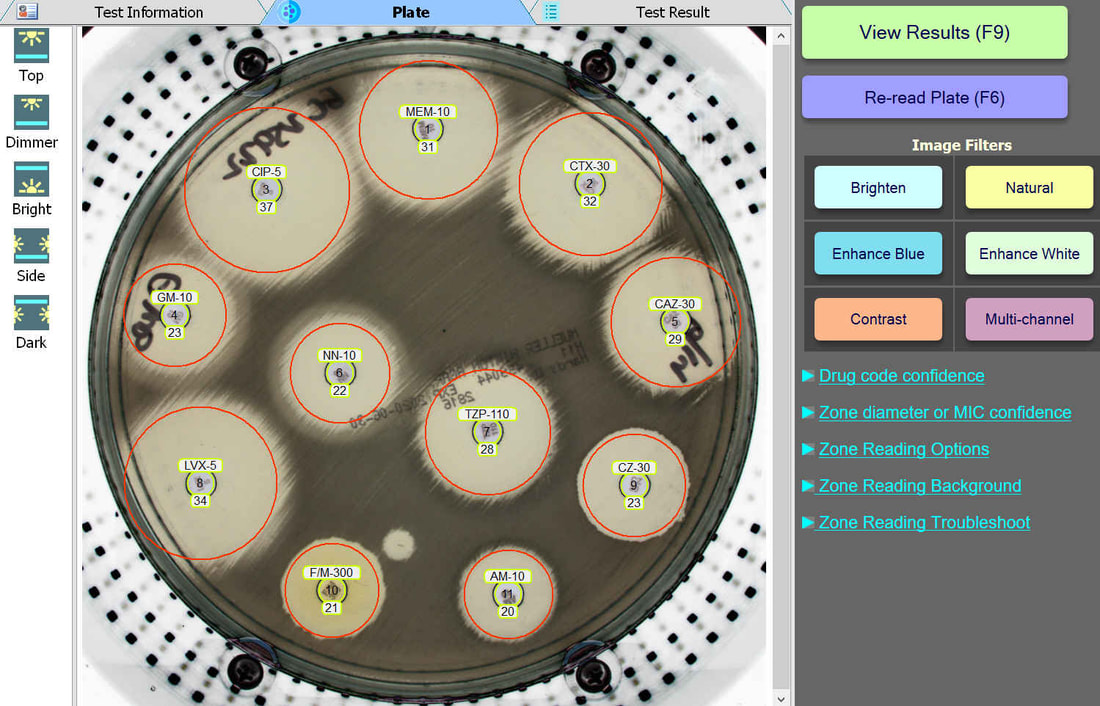
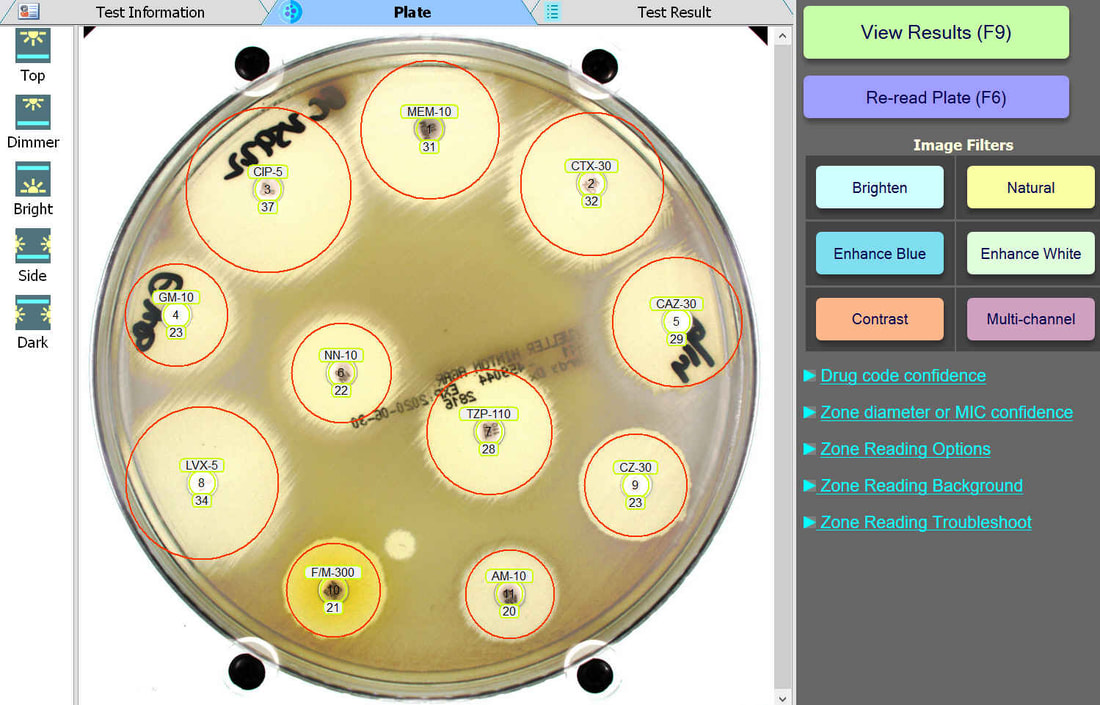
An enlarged plate image with antibiotic disks and zone diameter reading is displayed with the option to adjust on-screen by the user. Test results and high resolution images are saved automatically and may be printed or sent to your lab's LIS/LIMS.
BIOMIC V3 automates:
Quality Control & Inventory Management software is included with this module. Learn More >
|
Select the video above to view (1:02)
Select the icon on the bottom right of the video for full screen mode.
Click here to view all videos >
Select the icon on the bottom right of the video for full screen mode.
Click here to view all videos >
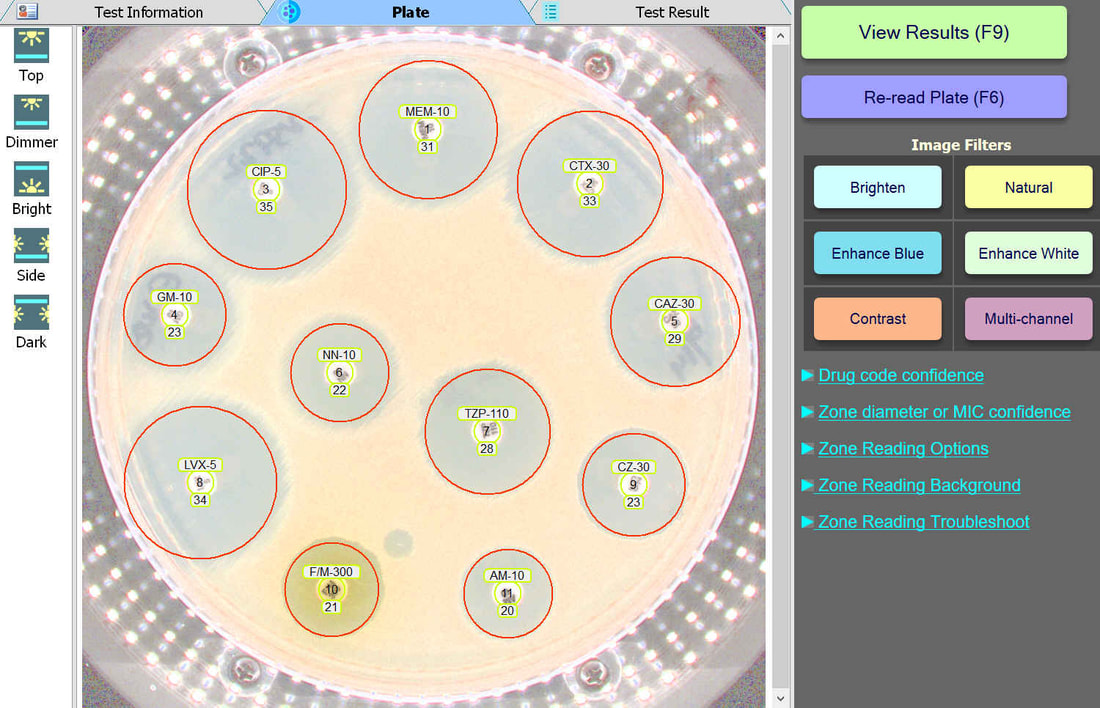
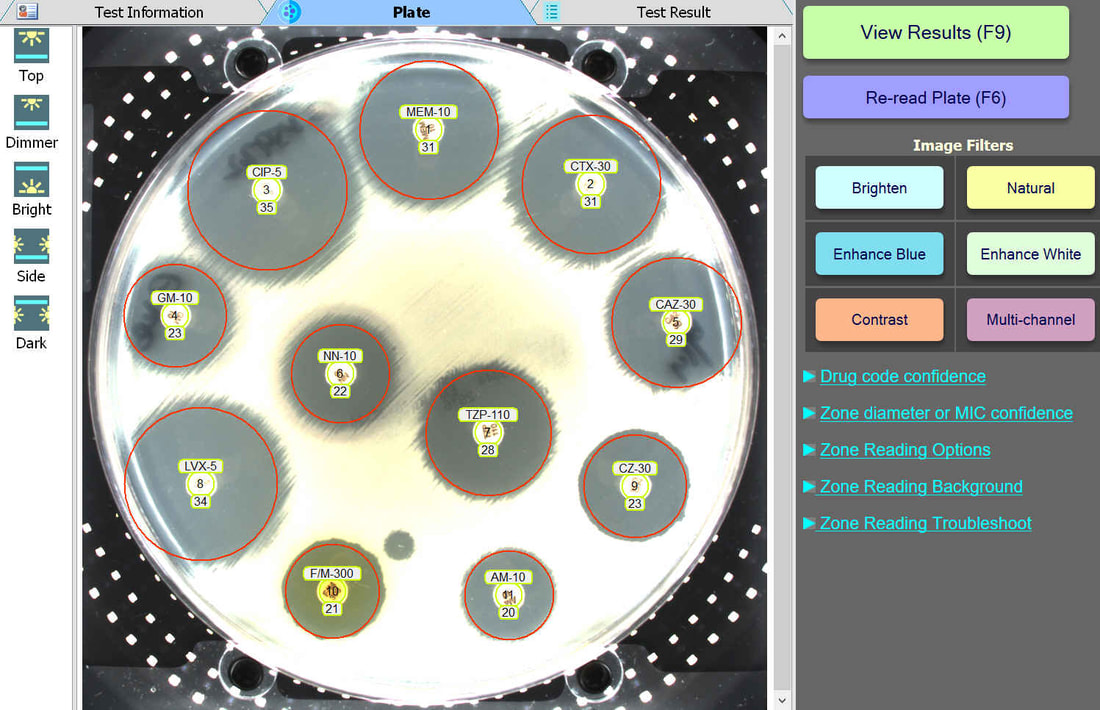
Plate Lighting
BIOMIC V3 customers may adjust the lighting source on-screen to help read plates with poor or weak growth. Each plate image is saved with multiple lighting options for future review.
Plate Image Filters
BIOMIC V3 customers may select from 6 image filters to help visualize difficult-to-read zones.
Resistance Monitoring & Antibiogram Reports
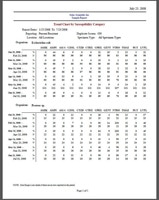
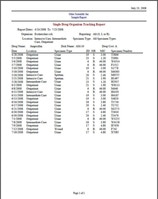
BIOMIC V3 provides antibiotic resistance monitoring following current CLSI or EUCAST guidelines. This includes a variety of antibiogram reports including Cumulative Susceptibility, Trend Chart/Graph by Susceptibility Category, Trend Graph by Zone Diameter, Distribution Graph by Zone Diameter, Location Specimen & Organism Count, Single-Drug Organism Tracking, Multi-Drug Organism Tracking, and more. Select the report below to enlarge image.
Automated Disk Code Recognition
BIOMIC V3 automatically recognizes antibiotic disk codes for BD and Oxoid disks. For example, BD disk code AM-10 is recognized as Ampicillin. If a BIOMIC V3 customer uses disks from a different manufacturer (example: Liofilchem), the customer would simply assign the disk positions on the plate during a one-time setup.
Rapid AST Direct from Positive Blood Culture
Rapid AST direct from positive blood culture guidelines from EUCAST (RAST) and CLSI are included with this module. Learn More >
Customer Testimonials - Disk Diffusion
"Standardized KB reading and QC results/image storage with BIOMIC V3"
"We use the BIOMIC V3 to read and interpret all of our Kirby Bauers (KB). We have a big population of Cystic Fibrosis patients in our hospital. We set up most of the isolates from this patient group by KB. BIOMIC V3 makes it much easier and faster to read and interpret these isolates. We sometimes setup up to 40 KB a day and BIOMIC V3 has really made it faster and easier to read. It has been very useful even for the mucoid or difficult to grow organisms. We may have to make a manual adjustment sometimes, but it is still faster and easier to read than manual reading. It also helps us standardize our readings. We also use BIOMIC V3 to read our KB QC. I also like the fact that it stores all the QC, patient readings and images. I can go back and check the image and the reading when needed. I also like and appreciate the great customer service and the yearly CLSI updates."
Tsigereda Tekle MT (ASCP)
Lead Clinical Lab Scientist
Johns Hopkins Hospital, Baltimore, MD, USA
BIOMIC V3 customer since 2010
“Excellent reading speed and precision with BIOMIC V3”
"BIOMIC V3 has been a great addition to our microbiology section. The speed and precision when reading the Kirby Bauer disc diffusion plates has been excellent. In addition to greater accuracy, the system saves a significant amount of time from reading the plates manually. The quality control software for the antibiotic discs, media and reagents is excellent and very user friendly. The support staff from BIOMIC are very helpful and available. We only wish we had adopted this technology sooner!"
Robert Gibson
Microbiology Section Head, Managing Director
New Hampshire Veterinary Diagnostic Lab
Durham, NH, USA
BIOMIC V3 customer since 2020
“BIOMIC V3: Keeping up with the latest CLSI guidelines”
We use BIOMIC V3 primarily on our busy HAI/ARLN bench. It is so simple to use, and the images are crystal clear. We use the instrument for reading our API 20E test strips and our Disk Diffusion by Kirby Bauer plates. The ability of the instrument to store and print out all the lot numbers and expiration dates for the vast about of antibiotics has been such a time saver. The speed which you can read and record both API 20E test strips and KB plates is a true time saver and helps prevent errors. We especially love how the manufacturer keeps up with the latest CLSI updates.
Sandra J. Bandstra
Clinical Microbiology Supervisor
South Carolina DHEC Public Health Laboratory, USA
BIOMIC V3 customer since 2018
“The BIOMIC auto-enhance feature has really improved the functionality”
"The BIOMIC auto-enhance feature has really improved the functionality...I have experienced your prompt response to customer suggestions before and I have been very impressed with the user-friendly interface of your software...Keep up the good work."
Brendan O'Reilly
Senior Medical Scientist, Dept. of Medical Microbiology
Cork University Hospital
Cork, Ireland
BIOMIC V3 customer since 2002
"Kirby-Bauer and Crystal ID panel automation for our vet lab"
"BIOMIC V3 has automated the reading of our Kirby-Bauer plates and Crystal ID Panels saving valuable tech time and helping to eliminate possible clerical errors. The BIOMIC software has incorporated CLSI guidelines which are necessary when interpreting and reporting results. The epidemiology reports are a valuable tool in monitoring drug resistance. The Giles support team and reliability of the system have exceeded our expectations."
Tina Elam
Microbiology Supervisor
Hagyard Equine Medical Institute, Lexington, KY, USA
BIOMIC V3 customer since 2006
"Accurate disc zone diameter reading & QC following EUCAST and CLSI guidelines"
"What I like about the BIOMIC V3 is its user friendliness. Simple to install and operate, with accurate reading of disc zone diameters. Result interpretations for both patients and QC can be based on either EUCAST or CLSI. Regular software upgrades are easy to download and install, while rapid technical support is always available."
Michael Huysmans
Senior Scientist
Microbiology Unit
Alfred Pathology Service, Victoria, Australia
BIOMIC V3 customer since 2010
"We use the BIOMIC V3 to read and interpret all of our Kirby Bauers (KB). We have a big population of Cystic Fibrosis patients in our hospital. We set up most of the isolates from this patient group by KB. BIOMIC V3 makes it much easier and faster to read and interpret these isolates. We sometimes setup up to 40 KB a day and BIOMIC V3 has really made it faster and easier to read. It has been very useful even for the mucoid or difficult to grow organisms. We may have to make a manual adjustment sometimes, but it is still faster and easier to read than manual reading. It also helps us standardize our readings. We also use BIOMIC V3 to read our KB QC. I also like the fact that it stores all the QC, patient readings and images. I can go back and check the image and the reading when needed. I also like and appreciate the great customer service and the yearly CLSI updates."
Tsigereda Tekle MT (ASCP)
Lead Clinical Lab Scientist
Johns Hopkins Hospital, Baltimore, MD, USA
BIOMIC V3 customer since 2010
“Excellent reading speed and precision with BIOMIC V3”
"BIOMIC V3 has been a great addition to our microbiology section. The speed and precision when reading the Kirby Bauer disc diffusion plates has been excellent. In addition to greater accuracy, the system saves a significant amount of time from reading the plates manually. The quality control software for the antibiotic discs, media and reagents is excellent and very user friendly. The support staff from BIOMIC are very helpful and available. We only wish we had adopted this technology sooner!"
Robert Gibson
Microbiology Section Head, Managing Director
New Hampshire Veterinary Diagnostic Lab
Durham, NH, USA
BIOMIC V3 customer since 2020
“BIOMIC V3: Keeping up with the latest CLSI guidelines”
We use BIOMIC V3 primarily on our busy HAI/ARLN bench. It is so simple to use, and the images are crystal clear. We use the instrument for reading our API 20E test strips and our Disk Diffusion by Kirby Bauer plates. The ability of the instrument to store and print out all the lot numbers and expiration dates for the vast about of antibiotics has been such a time saver. The speed which you can read and record both API 20E test strips and KB plates is a true time saver and helps prevent errors. We especially love how the manufacturer keeps up with the latest CLSI updates.
Sandra J. Bandstra
Clinical Microbiology Supervisor
South Carolina DHEC Public Health Laboratory, USA
BIOMIC V3 customer since 2018
“The BIOMIC auto-enhance feature has really improved the functionality”
"The BIOMIC auto-enhance feature has really improved the functionality...I have experienced your prompt response to customer suggestions before and I have been very impressed with the user-friendly interface of your software...Keep up the good work."
Brendan O'Reilly
Senior Medical Scientist, Dept. of Medical Microbiology
Cork University Hospital
Cork, Ireland
BIOMIC V3 customer since 2002
"Kirby-Bauer and Crystal ID panel automation for our vet lab"
"BIOMIC V3 has automated the reading of our Kirby-Bauer plates and Crystal ID Panels saving valuable tech time and helping to eliminate possible clerical errors. The BIOMIC software has incorporated CLSI guidelines which are necessary when interpreting and reporting results. The epidemiology reports are a valuable tool in monitoring drug resistance. The Giles support team and reliability of the system have exceeded our expectations."
Tina Elam
Microbiology Supervisor
Hagyard Equine Medical Institute, Lexington, KY, USA
BIOMIC V3 customer since 2006
"Accurate disc zone diameter reading & QC following EUCAST and CLSI guidelines"
"What I like about the BIOMIC V3 is its user friendliness. Simple to install and operate, with accurate reading of disc zone diameters. Result interpretations for both patients and QC can be based on either EUCAST or CLSI. Regular software upgrades are easy to download and install, while rapid technical support is always available."
Michael Huysmans
Senior Scientist
Microbiology Unit
Alfred Pathology Service, Victoria, Australia
BIOMIC V3 customer since 2010